  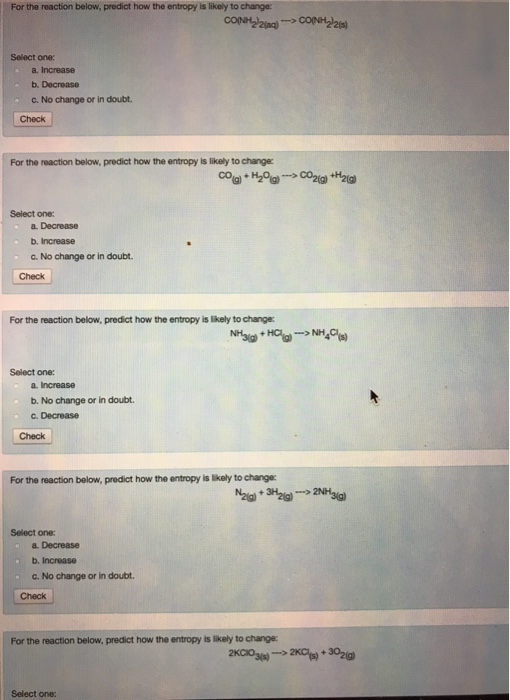
Moving in the direction of greater disorder in an isolated system is one of the two forces that drive change. The preferred direction in nature is toward maximum entropy. The second law of thermodynamics states that the total entropy of a chemical system and that of its surroundings always increases if the chemical or physical change is spontaneous. This state is one of near maximum disorder and near maximum entropy. 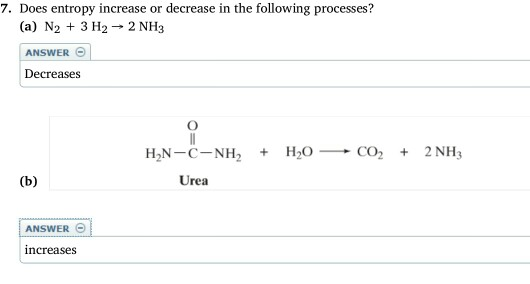
Each gas particle moves more or less independently of the other particles. This looser arrangement of ions in molten sodium chloride shows that the liquid state displays an increased disorder than the solid state, so it is of higher entropy.Ī gas has even greater disorder than a liquid because its constituent molecules or atoms are no longer constrained to be adjacent to each other. No longer would each ion be surrounded by precisely 6 ions of the opposite charge. Each Na +, therefore, will be adjacent to as many Cl – anions as possible, and each Cl – will tend to be surrounded by Na + cations. They will move relative to each other, subject only to the constraint of electrostatic attraction and repulsion. When solid NaCl is heated to 801☌, it melts, and the ions are no longer fixed in a simple geometric pattern. The regular, repetitive structure has a high degree of order and low entropy or low disorder. If you examine the NaCl crystal structure closely, you will see that each Cl – is surrounded by 6 Na +, and each Na + is surrounded by 6 Cl –. Solid sodium chloride has a crystalline structure in which the cations and anions alternate in a repeating pattern. As an illustration, compare the arrangements of Na + and Cl – ions in both solid and liquid sodium chloride. The second law of thermodynamics involves entropy, which for our purposes is a statistical measure of the degree of disorder in a chemical system. An exothermic reaction releasing heat to the surroundings, therefore, is accompanied by a decrease in internal energy, whereas an endothermic reaction has a concomitant increase in internal energy. The first law of thermodynamics also tells you that if no work is done on or by the sample-that is, pressure and volume are held constant-any heat flow is counterbalanced by a change in internal energy. Expansion against a confining pressure reduces the internal energy, whereas external compression of the system increases the internal energy. If no heat flows in or out of a sample of matter, any external work done on or by the sample is precisely offset by the opposite change in internal energy. The internal energy is sometimes called chemical energy because it is the consequence of all the motions of particles and forces between particles: molecules, atoms, nucleons, and electrons. The three major forms of energy for chemical purposes are the internal energy of each substance, the external work due to changes in pressure or volume, and the exchange of heat with the surroundings. The first law of thermodynamics asserts that energy is conserved during any process. Begin by reviewing the two main laws of the field. 
Many powerful calculations in thermodynamics are based on a few fundamental principles, which are called the laws of
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
